 Note that the percentages appear small compared to the 5% level of baking powder suggested because baking powder contains both an acid agent and a leavening agent. Table 11 provides the recommended amounts of baking soda for different products. Cookies, for example, with high levels of fat and sugar, do not require much, if any, leavening. The level of baking soda depends on the nature of the product and on the other ingredients in the formula. On the other hand, the reddish, coarse, open-grained crumb in devil’s food cake is the result of using baking soda as the principal leavening agent. A darker and richer chocolate colour is produced if the acid level is sufficient to release all the carbon dioxide gas. This means more baking powder and less baking soda is used.īaking soda in a chocolate mix not only counteracts the acid content in the baked cake but also improves the grain and colour of the cake. In the Dutch process, some of the acid in the cocoa is already neutralized, and there is less left for the release of gas in the mix. However, it is important to know whether the cocoa powder you are using is natural or treated by the Dutch process. In chocolate cakes, baking soda is used in conjunction with baking powder to keep the pH at a desirable level. In spiced honey cookies and gingerbread, baking soda is used alone to give them quick colour during baking and yet keep the products soft. The darker colour of the crumb found on the bottom half of a cake or muffins is caused by the partial dehydration of the batter that is heated first during baking. Mixes and doughs leavened with baking soda must be handled without delay, or the release of the gas may be almost exhausted before the product reaches the oven. 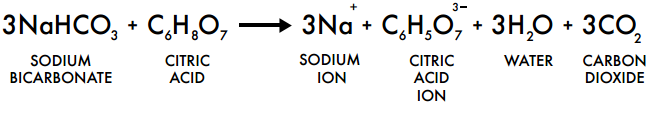
Once the soda neutralizes the acid, the dough or batter quickly becomes alkaline and the release of gas is reduced. The gas evolves very fast at the beginning of baking when the pH level is still on the acidic side (pH of around 5 to 6). If an excessive amount of baking soda is used in a cake batter without the presence of sufficient acid, the normally white cake crumb will have a yellowish-brown colour and a strong undesirable smell of soda. Examples are:įor this reason, some of the mixes contain baking powder only while others contain a combination of baking powder and baking soda. Slightly acidic ingredients provide the mix with some of the necessary acids for the release of carbon dioxide gas. If it is moistened and heated in the presence of sufficient acid, it will release twice as much gas as if it is moistened and heated without the presence of an acid. some cakes).When sodium bicarbonate (baking soda) is moistened and heated, it releases carbon dioxide gas. Baking soda won't work in recipes that call for baking powder and have no acidic ingredient (e.g. They are somewhat interchangeable in recipes - but they have different taste - baking soda needs an acid to react and to balance out the bitter, basic taste, whereas baking powder has a more neutral taste (since it already contains an acid and a base). This reaction happens immediately upon mixing ingredients, so you need to bake quickly after mixing for maximum rise.Baking powder is sodium bicarbonate, mixed with an acidifying agent (cream of tarter) and a drying agent (starch). When baking soda is mixed with moisture and an acidic ingredient (e.g., yogurt, chocolate, buttermilk, and honey), the resulting chemical reaction produces bubbles of carbon dioxide that expand when cooking in the oven, causing baked goods to rise. (4.2) N a 2 C O 3 H 2 O + 2 H X 2 N a X + C O 2 + 2 H 2 O Sodium carbonate + acid salt + carbon dioxide + water 0.74 g from Formula 1 a. 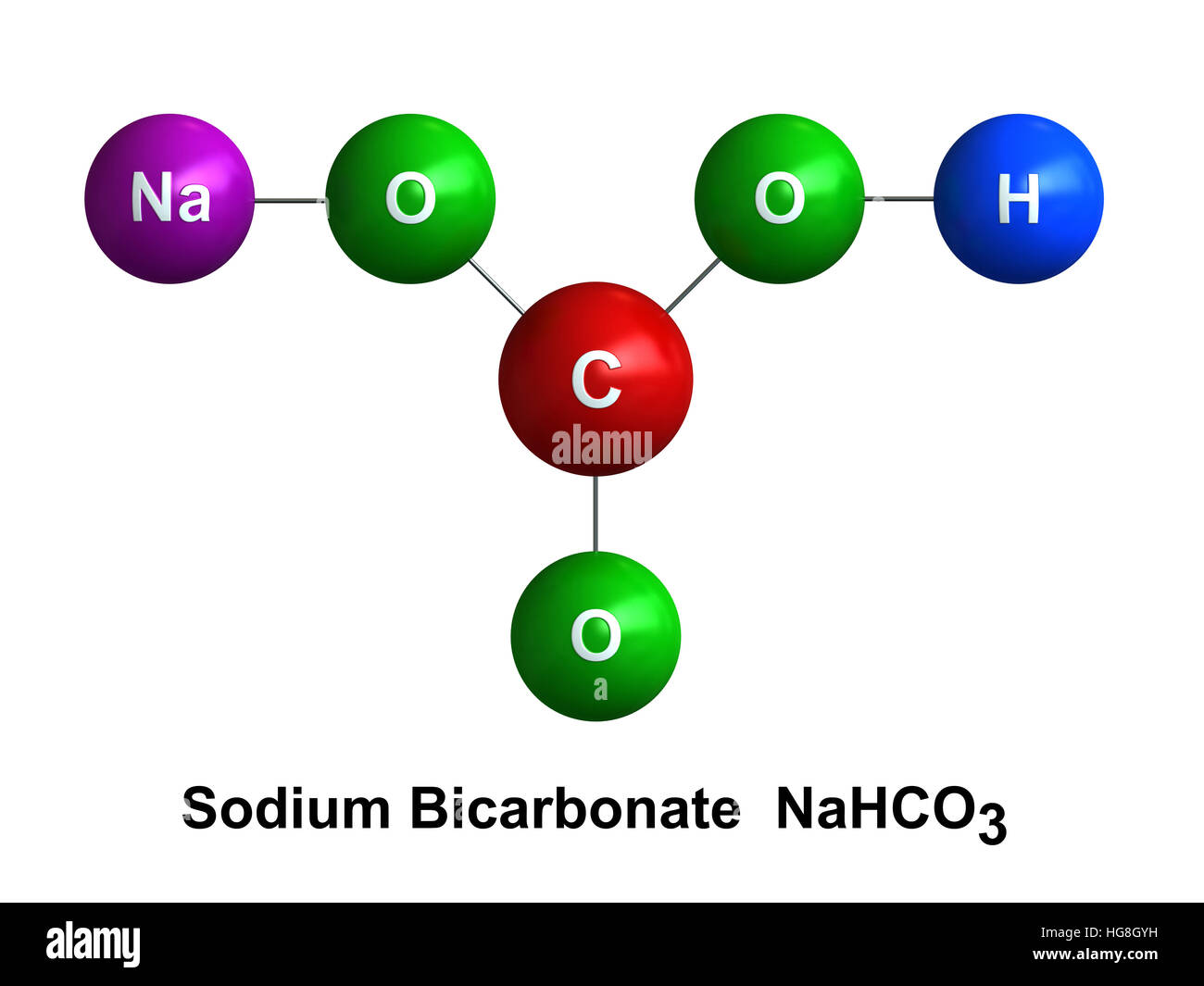
CO 2 can then come out of solution, becoming carbon dioxide gas, which forms bubbles, thereby causing the bread to rise.īaking soda is sodium bicarbonate (a chemically basic compound). H 2CO 3 has a tendency to dissociate into carbon dioxide, CO 2, and water, H 2O. In the presence of acid, H+, the bicarbonate ion combines with the acid protons to become hydrogen carbonate or carbonic acid, H 2CO 3. That dissolves in water to become Na+ plus HCO 3. How does baking soda work? What properties in it make dough rise?īaking soda is sodium bicarbonate, NaHCO 3. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
